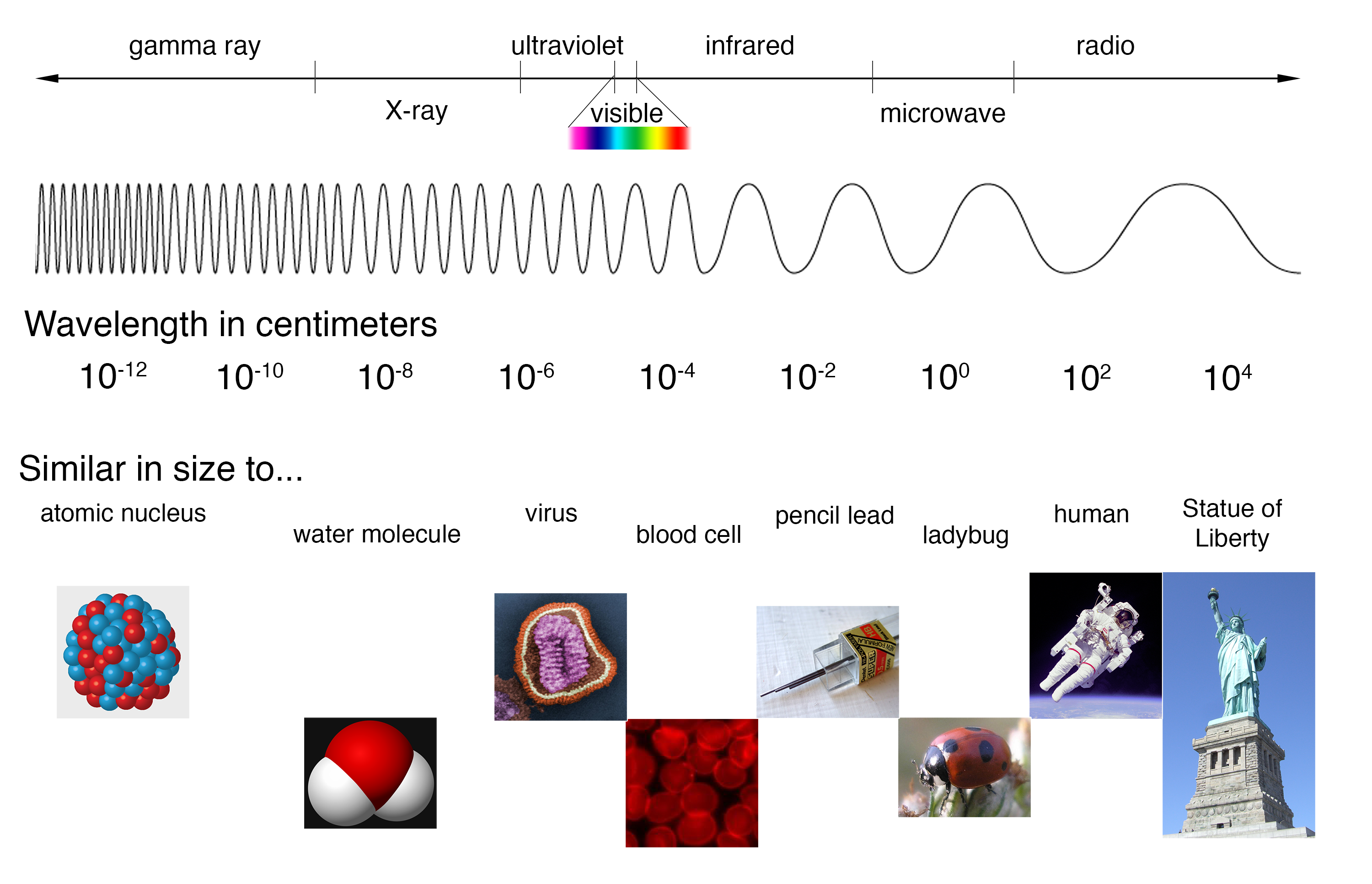
Visible Spectra of the Elements

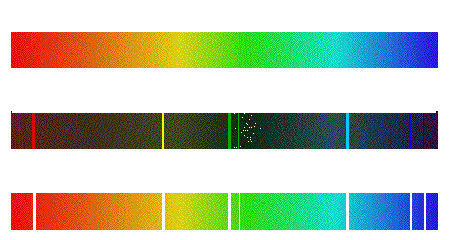
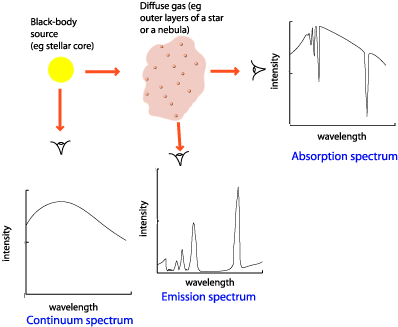
Difference Between Continuous Spectrum and Line Spectrum
Why do atoms of certain elements emit light of a specific color when heated? - Quora

Helium emission and absorption spectra - Stock Image - C025/8080 - Science Photo Library

ektalks: What is Light? All You Need to Know - A Community Education Feature

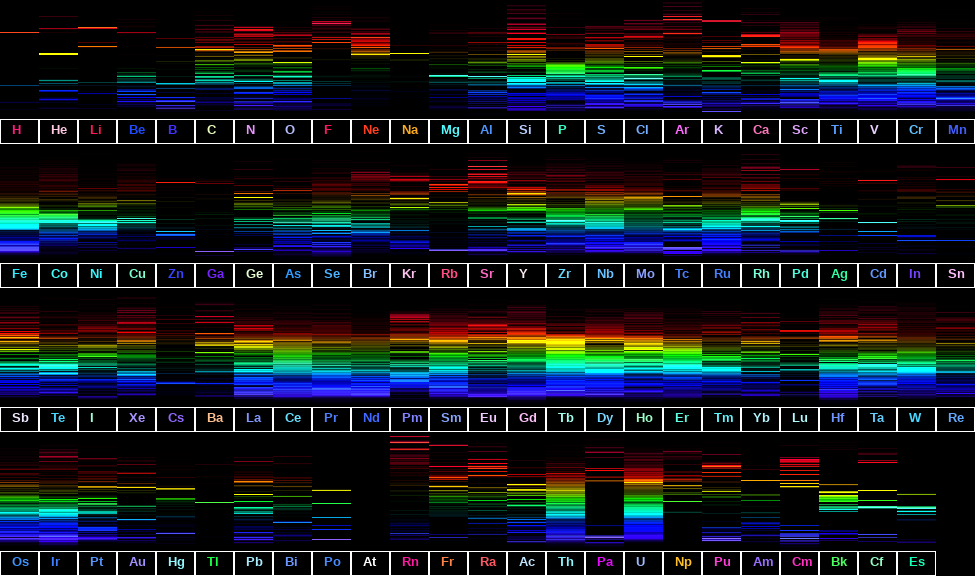
Spectral Lines of Elements in the Periodic Table / Mario Delgado

This is what the Periodic Table of the Elements looks like with the elements represented by th…
Why does potassium have a shorter wavelength than barium when conducting a flame test? - Quora

Frequent variations in tandem repeats pSc200 and pSc119.2 cause rapid chromosome evolution of open-pollinated rye
If potassium gives violet/lilac color in flame test, then why at all places wavelength of this color given is 766.5nm it is very high wavelength which lies in red region ,not in

Elemental Spectra: Unveiling the Colors of the Elements

White Light, Definition, Wavelength & Spectrum - Lesson

Spectroscopy: Reading the Rainbow